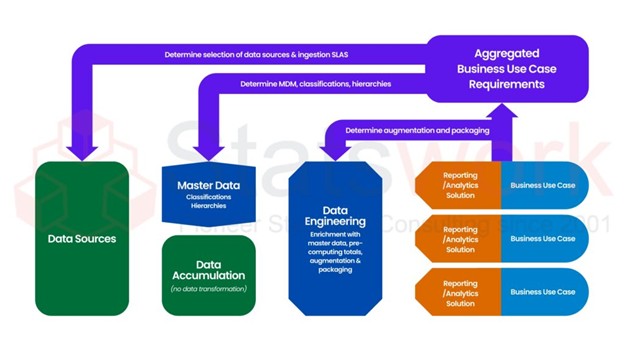
HIS Methodology Assessment and Enhancement
We provide a complete range of support for enhancing HIS methodologies through reviewing and revising them to maximize performance, compliance and workflow efficiency. Our team of experts collaborate with healthcare entities to improve system efficiencies, increase data quality, and improve patient care.
Optimizing Methodologies for Health Information Systems
Review and revision of methodologies in Health Information Systems is important to keep the system compliant, to enhance operational performance of the system and to improve Data Integrity within the HIS. The process of methodology revision generally requires both a clinical perspective and technical expertise; therefore, it is essential to not only improve System Performance but to also be able to align the Workflow with Regulatory Compliance. We provide a full-service solution to reviewing and revising HIS Methodologies. We will evaluate what you currently have, identify areas of opportunity (Gaps) with your existing workflow, and develop improvements to your current methodologies.

Our Subject Matter Experts will work with your team through all stages, from Initial Evaluation and Gaps Analysis through Implementation, Testing, and Documentation to ensure that your HIS is compliant, scalable, and functioning efficiently.
Using the best practices within the industry for System Design and Data Management, we provide Health Care Organizations with the ability to streamline their Workflow Process, and increase the ability for Data Interoperability and provide the ability to Improve Reporting Accuracy. We also empower your team to optimize their methodology, make Data-Driven Decisions and provide Quality Patient Care via Digital Systems.

Research-Evidence-based Approach
Statswork provides an evidence-based methodology review/revision of your proposal based on the current best practices of methodological research.

Statistical Rigor and Quality Assurance
Using highly trained and knowledgeable research staff, Statswork reviews/edits your methodology to ensure it's free of bias, statistically valid, and adheres to the International Standard for Reporting Results.

Transparency and Scientific Integrity
Statswork operates according to internationally accepted principles of transparency in the revision process and the ability to reproduce the results of your research, maximizing the impact of your research and providing measurable ROI.
Evaluating and Improving Study Methodology
We conduct a methodical evaluation of your study methodology and identify opportunities for improvement to enhance study design and analysis. Our goal is to increase the validity, reliability, and accuracy of your study.

Addressing Potential Sources of Bias
We work with you to identify potential sources of bias in your study design and propose strategies to address them, thereby reducing the likelihood of unwanted bias and increasing the reliability and scientific validity of your results.

Reviewing Compliance with Ethical and Regulatory Standards
We ensure that your study methodology follows all applicable ethical and regulatory standards (e.g. ICH-GCP, FDA, EMA) to facilitate the smoothest possible approval process and to guarantee compliance with all relevant regulatory agencies.

Making the Methodology of Your Study Reproducible and Transparent
We help you create a transparent and reproducible methodology so that others can repeat your research. This provides scientific integrity and builds trust in your research results.

Our Sector
In the Healthcare, Pharmaceutical and Research Industries, Statswork specializes in the rewriting and/or reviewing of methodologies. We help increase the quality of Study Designs, Process the Data Better, and Ensure Compliance with Regulations; thus, enhancing the Integrity of Research and Improving the Reliability and Reproducibility of Results.
Refining Study Design and Analysis: Effective methods are imperative to conduct rigorous and scientifically valid research that meets regulatory requirements and research objectives. Therefore, we refine the method you use to conduct the analysis to ensure that it meets the above criteria and generates reliable findings that can be disseminated through peer-reviewed publication.
Methods that are Thoroughly Documented and Follow Best Practices: We take all best practices into account when making revisions to your method. By doing this, we help to ensure that your method is compliant with international standards, is properly documented, and can be defended when challenged.
Increasing Productivity and Quality of Research Outcomes: With an optimized method, we help facilitate a more streamlined approach to executing your research. Because of this optimization, you will experience fewer mistakes, improved productivity, reproducible results, and ultimately better-quality research outcomes.




1. Evaluation of Methodology
We assess and enhance the design of study to ensure validity; clarity; scientific integrity; conformance with accepted standards of research worldwide (e.g., ICH GCP, CONSORT).

2. Optimization of Methodology
Expertise is applied to Methodology Development for optimal design of study; identification of data flow/processes; development of Analyses; and preparation of Study Implementation Documentation (SID); ensuring a comprehensive plan for implementing the study. The resultant study will be reproducible, and the results will be valid and interpretable.

3. Regulatory Compliance and Ethical Standards
Our methodology is designed to include all regulatory and ethical guidelines in order to minimize bias and ensure conformance with all Research Regulatory Standards throughout the world.
4. Review and Enhancement of Methodology
An in-depth review and enhancement of study design will be conducted for clarity, feasibility, and methodological integrity. As a result, the credibility and validity of the research will be enhanced.

5.Strategic Guidance During Implementation
Ongoing strategic direction will be provided to your study team throughout the entire study process, assuring that study outcomes will be achieved with confidence, transparency and reproducibility.





Methodology review and revision is the process of evaluating, improving, and refining research methods, study design, sampling techniques, and analytical approaches to ensure research accuracy and validity.
Methodology revision helps improve research reliability, reduce errors, strengthen study design, and ensure that the research approach aligns with academic and scientific standards.
Statswork reviews research objectives, study design, sampling methods, statistical approaches, data collection procedures, and analytical frameworks to improve overall research quality.
PhD scholars, academic researchers, healthcare professionals, clinical researchers, and business analysts can benefit from methodology review and revision services.
Yes, a well-structured and validated methodology increases the chances of journal acceptance, research approval, and successful thesis or dissertation completion.
Yes, Statswork provides methodology review support for clinical trials, biostatistics studies, healthcare research, epidemiology, and pharmaceutical research projects.
Begin dealing with the shopping behaviour data to convert more shoppers to buyers and personalize the buyer journey today!