What is Real World Evidence?
- Home
- Insights
- Article
- What is Real World Evidence?
Qualitative Research Service
News & Trends
Recommended Reads

Data Collection
As the data collection methods have extreme influence over the validity of the research outcomes, it is considered as the crucial aspect of the studies
- 1. Introduction
- 2. DeepHealth’s Diagnostic Suite™: Revolutionizing Radiology Workflows
- 3. Key Features
- 4. AI Impact on National Screening Programs
- 5. SmartMammo™: Enhancing Breast Cancer Screening
- 6. DeepHealth AI Use Cases Across Specialties
- 7. Strategic Collaborations and Ecosystem Expansion
- 8. Impact and Adoption of DeepHealth’s AI Solutions
- 9. Conclusion: The Future of Radiology with AI
- 10. References
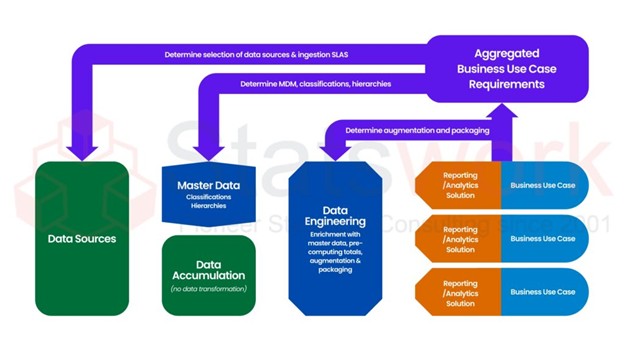
Insights from clinical examination of Real world evidence (RWE), which is the result of the collection of evidence outside of a clinical trial, will inform about how a given treatment or intervention is likely to perform when given in routine clinical practice and can help establish the effectiveness, safety and long term effects of an intervention on patients with multiple conditions.[1]
Real-World Data: Major Origins and Sources
Real-world evidence is derived from several healthcare data sources, such as:
- Electronic Health Records (EHRs) that hold longitudinal patient records (what happened when)
- Claims and billing data documenting diagnoses, procedures and costs
- Disease registries based on the diseases or types of therapy used
- Patient Reported Outcomes (PROs) capturing the actual experience of treatment
- Digital Health Data is captured through wearables, mobile health applications, and other forms.
All these data sources together create an ongoing and comprehensive view of patient care and results.[2]
How Real-World Evidence Improves Healthcare Outcomes
Real-World Evidence is valuable to health care stakeholders.
- Treatment Effectiveness: Evaluates therapy’s performance beyond clinical trial control.
- Safety Monitoring: Early detection of adverse events and long-term risks associated with therapy.
- Informed Decision Making: Used by clinicians, payers and policymakers in their decision making.
- Patient Representation: Includes diverse populations that are not typically included in clinical trials.
All the above enhance clinical decision-making in the real world.[3]
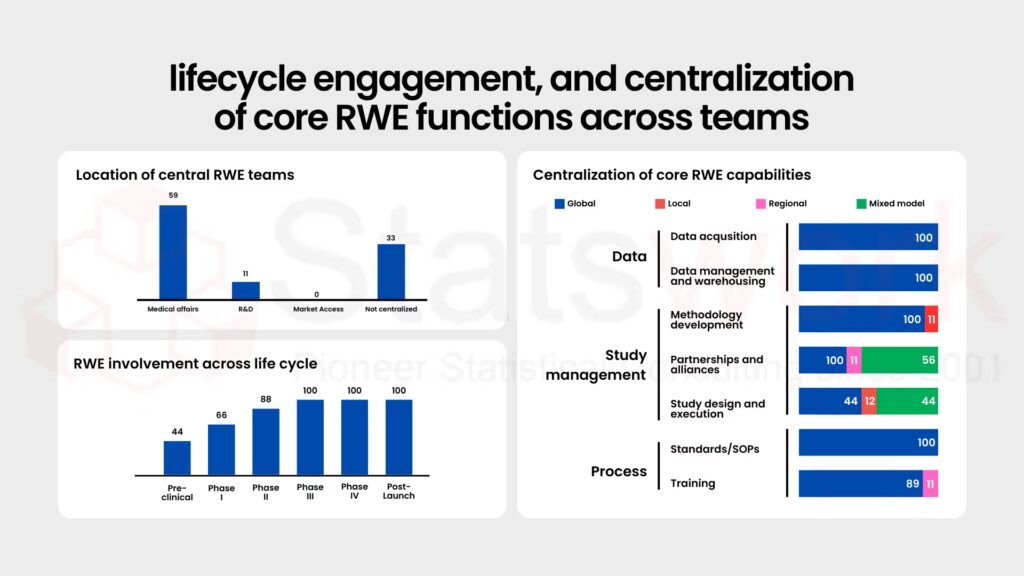
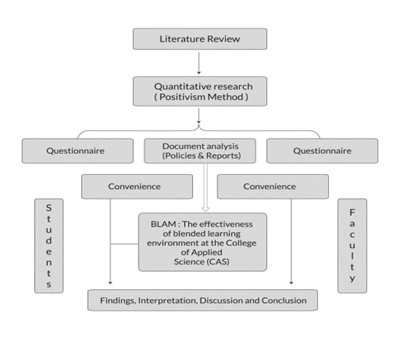
Fig. 1 shows the distribution, lifecycle engagement, and centralization of core RWE functions across teams.
Clinical Trials vs Real-World Evidence
Aspect | Clinical Trials | Real-World Evidence |
Study Setting | Controlled research environment | Routine clinical practice |
Patient Population | Selective participants | Diverse and inclusive patients |
Data Collection | Fixed, structured protocols | Observational and ongoing |
Outcome Focus | Short-term endpoints | Long-term, real-life outcomes |
Generalizability | Limited | High and broadly applicable |
Role of RWE in Regulation, Access, and Analytics
Healthcare Systems are Supported through real-world evidence, as shown below:
- Regulatory Support: Supporting Post-market Surveillance and Label Expansions for Regulatory Agencies
- Health Economics & Outcomes Research (HEOR): Aid in Reimbursement Decision Making and Value-Based Healthcare Evaluations
- Data Analytics: Increase Quality of Care and Improve Population Health via Advanced Analytics
- Clinical Research Support: Support Clinical Research with Continued Regulatory Compliance.[4]
Real-World Evidence in Patient-centred Outcome Measurement
This Approaches highlights how the RWE can demonstrate which outcomes matter to patients, how they are achieved through real-life effectiveness, real-life experience, and real-life value.
- Clinical Outcome Insights: By measuring the results of a treatment over an extended period, RWE can evaluate long-term treatment effectiveness, as well as how diseases progress and their durability as compared to clinical trial settings and different patient populations.
- Humanistic and Quality-of-Life Outcomes: Patient self-reporting provides valuable information for measuring patient-reported symptoms, functional status, and overall health and well-being in standard healthcare practice.
- Economic and Resource Utilization Outcomes: RWE can be an important part of determining the cost-effectiveness of healthcare services and forming decisions based on value of those services.[5]
Conclusion
In the modern healthcare setting, Real World Evidence (RWE) will play an important role in taking real world clinical data and turning it into valuable information we can act on. Improved safety surveillance, wider patient representation and a bigger role for data in making decisions will enhance how we deliver healthcare and support what we do today through traditional clinical research methods.
Transform real-world data into real-world impact with StatsWork Biostatistics!
Reference
- Sherman, R. E., Anderson, S. A., Dal Pan, G. J., Gray, G. W., Gross, T., Hunter, N. L., … & Califf, R. M. (2016). Real-world evidence—what is it and what can it tell us. N Engl J Med, 375(23), 2293-2297. https://d1wqtxts1xzle7.cloudfront.net/80939023/4049b0a52404ade5913a3669110006f3a1b1-libre.pdf?1645054356=&response-content-
- O’Leary, C. P., & Cavender, M. A. (2020). Emerging opportunities to harness real world data: an introduction to data sources, concepts, and applications. Diabetes, Obesity and Metabolism, 22, 3-12. https://dom-pubs.onlinelibrary.wiley.com/doi/full/10.1111/dom.13948
- Katkade, V. B., Sanders, K. N., & Zou, K. H. (2018). Real world data: an opportunity to supplement existing evidence for the use of long-established medicines in health care decision making. Journal of multidisciplinary healthcare, 295-304. https://www.tandfonline.com/doi/full/10.2147/JMDH.S160029
- McNair, D., Lumpkin, M., Kern, S., & Hartman, D. (2022). Use of RWE to inform regulatory, public health policy, and intervention priorities for the developing world. Clinical Pharmacology & Therapeutics, 111(1), 44-51. https://ascpt.onlinelibrary.wiley.com/doi/full/10.1002/cpt.2449
- Justo, N., Espinoza, M. A., Ratto, B., Nicholson, M., Rosselli, D., Ovcinnikova, O., … & Drummond, M. F. (2019). Real-world evidence in healthcare decision making: global trends and case studies from Latin America. Value in Health, 22(6), 739-749. https://www.sciencedirect.com/science/article/pii/S1098301519300774