What is Clinical Trial Programming in Simple Terms?
- Home
- Insights
- Article
- What is Clinical Trial Programming in Simple Terms?
Qualitative Research Service
News & Trends
Recommended Reads

Data Collection
As the data collection methods have extreme influence over the validity of the research outcomes, it is considered as the crucial aspect of the studies
- 1. Introduction
- 2. DeepHealth’s Diagnostic Suite™: Revolutionizing Radiology Workflows
- 3. Key Features
- 4. AI Impact on National Screening Programs
- 5. SmartMammo™: Enhancing Breast Cancer Screening
- 6. DeepHealth AI Use Cases Across Specialties
- 7. Strategic Collaborations and Ecosystem Expansion
- 8. Impact and Adoption of DeepHealth’s AI Solutions
- 9. Conclusion: The Future of Radiology with AI
- 10. References
Clinical trial programming is one aspect of clinical research in which the programmer uses computer programs (structured programming and analytical workflows) to produce useful, accurate and regulatory-ready outputs from the raw data generated by the clinical trial. Clinical trial programming is critical throughout the entire clinical trial life cycle.[1]
Role Across the Clinical Trial Lifecycle
Programming clinical trials provides study support throughout the life cycle of study.
- Through simulated and mock outputs, clinical programming trials can assist with assessing protocol feasibility based on historical data.
- Programming clinical trials helps facilitate interim analysis of safety and effectiveness, providing critical information as studies progress.
- Programming clinical trials also enables statistical analysis of study results for Clinical Study Reports (CSRs).
- Finally, programming clinical trials guarantees the consistency of clinical trial data from study initiation through submission to the appropriate regulatory agency.[1]
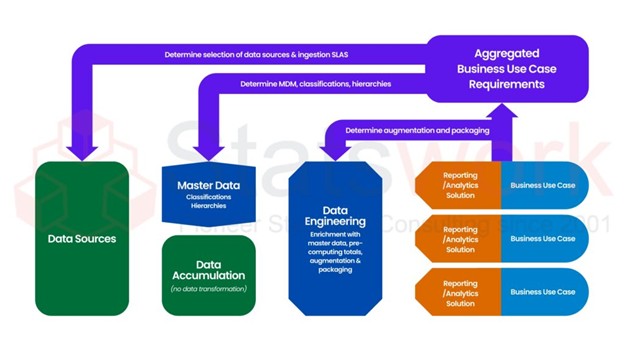
Data Integration and Standardization
The consolidation of data from multiple data sources into standardized data formats is the responsibility of a programmer.
- A programmer integrates data from EDC systems, laboratory tests, imaging, and other external vendors into one complete set of data.
- A programmer converts the raw data into CDISC compliant SDTM datasets.
- A programmer derives analysis-ready ADaM datasets that can be used for statistical analysis.
- A programmer maintains traceability from the original data source to the final output.[2]
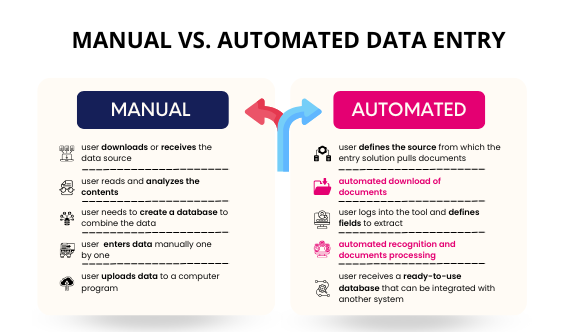
Automation and Efficiency in Programming
In modern clinical trial programming automation is playing an increasing role to speed up and increase the precision of the clinical trials.
- Automates & creates code that re-used, macros & functions/libraries.
- Automates data cleansing, validation, & reporting process.
- Reduces the opportunities for human error & eliminates duplication in programming.
- Makes it possible to complete trials faster & still deliver accurate, high-quality data.[3]
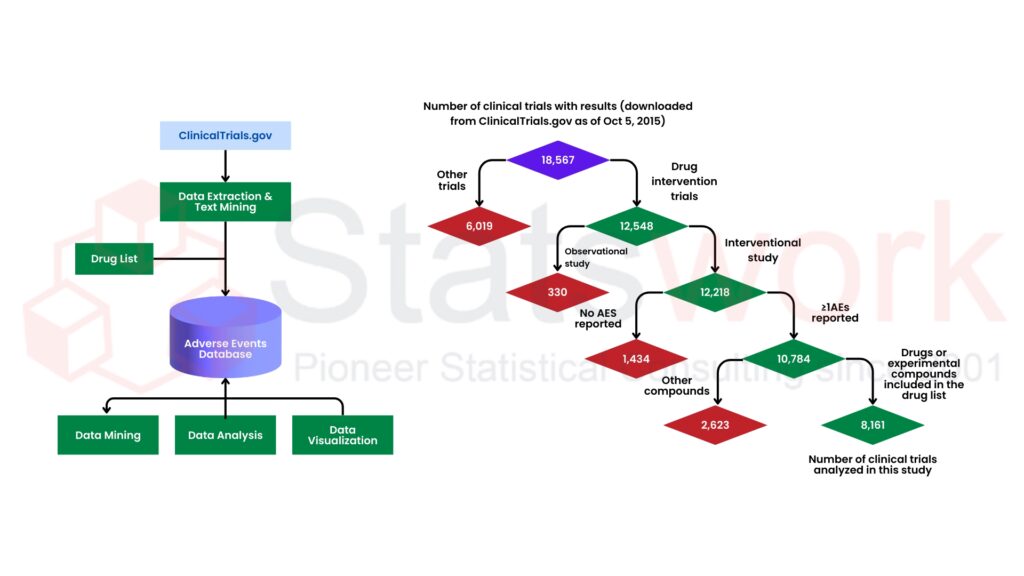
Fig 1 shows the workflow of clinical trial data extraction, adverse event database creation, and drug intervention trial selection.
Quality Control and Validation
Clinical trial programmers have a primary responsibility to maintain data accuracy and reliability.
- Independent programming validation and double programming are performed by clinical trial programmers.
- Clinical trial programmers identify discrepancies, missing data, and protocol deviations.
- Programming logic and validation results are documented by clinical trial programmers.
- Clinical trial programmers support audit readiness and meet inspection requirements.[4]
Regulatory Submission Support
Programming for clinical trials is critical for obtaining a successful regulatory response from regulators/approvals.
- Preparing datasets/submission-ready and define.xml files for regulatory submissions.
- Compliant TLFs (tables, listings and figures) that meet the requirements or expectations of the relevant authorities.
- Transparency, reproducibility and data integrity for accurate document and auditability.
- Guarantees a timely and appropriate response to any regulatory request regarding specific data or other issues.[4]
Emerging Technologies and Tools
As the field continues to develop using innovative technology and more sophisticated programming methodologies.
- Uses R and Python as well as traditional environments such as Commercial SAS
- Utilizes visualization technologies to assist with exploration data analysis
- Leverages the use of data processing on Cloud platforms to facilitate scalable solutions
- Aligns with the trend toward regulatory acceptance of open-source software.[5]
To summarize, programming in clinical research is the infrastructure that connects unstructured clinical data with scientific evidence. Mainly by utilizing the processes of standardizing, automating and validating the data as well as ensuring that we have met all regulatory requirements, we are able to produce results that are scientifically sound and can ultimately be used to make decisions by both scientists and government officials.
Transform complex clinical data into clear, confident decisions with StatsWork’s innovative Biostatistics expertise.
Reference
- Saghaei, M. (2011). An overview of randomization and minimization programs for randomized clinical trials. Journal of Medical Signals & Sensors, 1(1), 55-61. https://journals.lww.com/jmss/fulltext/2011/01010/An_Overview_of_Randomization
- Le Sueur, H., Bruce, I. N., Geifman, N., & Masterplans Consortium. (2020). The challenges in data integration–heterogeneity and complexity in clinical trials and patient registries of Systemic Lupus Erythematosus. BMC medical research methodology, 20(1), 164. https://link.springer.com/article/10.1186/s12874-020-01057-0
- Penberthy, L., Brown, R., Puma, F., & Dahman, B. (2010). Automated matching software for clinical trials eligibility: measuring efficiency and flexibility. Contemporary clinical trials, 31(3), 207-217. https://www.sciencedirect.com/science/article/abs/pii/S1551714410000352
- DeVito, N. J., Bacon, S., & Goldacre, B. (2020). Compliance with legal requirement to report clinical trial results on ClinicalTrials. gov: a cohort study. The Lancet, 395(10221), 361-369. https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(19)33220-9/abstract?ved=2ahUKEwjvnq3v4oznAhUINOwKHVo3DYYQxfQBMAB6BAgCEAE&sa=X
- Gill, S. K., Christopher, A. F., Gupta, V., & Bansal, P. (2016). Emerging role of bioinformatics tools and software in evolution of clinical research. Perspectives in clinical research, 7(3), 115-122. https://journals.lww.com/picp/fulltext/2016/07030/Emerging_role_of_bioinformatics_tools