What is Clinical study Report Generation?
- Home
- Insights
- Article
- What is Clinical study Report Generation?
Qualitative Research Service
News & Trends
Recommended Reads

Data Collection
As the data collection methods have extreme influence over the validity of the research outcomes, it is considered as the crucial aspect of the studies
- 1. Introduction
- 2. DeepHealth’s Diagnostic Suite™: Revolutionizing Radiology Workflows
- 3. Key Features
- 4. AI Impact on National Screening Programs
- 5. SmartMammo™: Enhancing Breast Cancer Screening
- 6. DeepHealth AI Use Cases Across Specialties
- 7. Strategic Collaborations and Ecosystem Expansion
- 8. Impact and Adoption of DeepHealth’s AI Solutions
- 9. Conclusion: The Future of Radiology with AI
- 10. References
CSR Generation is a structured, legally required method of developing scientific reports to describe how clinical trials were designed, performed, analyzed, and produced results. A Clinical study Report (CSR) is an essential element of any legal submission to the appropriate regulatory authorities and provides the final piece of evidence for establishing that a new drug or device is safe and effective.
Purpose of Clinical Study Report
- To produce a full, precise, and open summary of a clinical trial for regulatory agencies (FDA, EMA, MHRA, and CDSCO).
- To facilitate an independent assessment of both the risks/benefits of a clinical trial as well as its scientific validity, and the integrity of the underlying data.
- To support regulatory decision-making and eventual market authorization approval by demonstrating adherence to pertinent guidelines.
What a Clinical Study Report Includes
The Clinical Study Report (CSR) will be created following the ICH E3 Guidelines to ensure that all portions are consistent and understandable. Components of a CSR include:
Study overview: The objective(s), design, methods, and treatments of the clinical trial.
Patient demographics: The subjects enrolled into the trial, their disposition following completion of the trial, and their baseline characteristics prior to entering the trial.
Efficacy results: The analysis of the primary and secondary endpoints.
Safety analysis: Adverse events (AEs), serious adverse events (SAEs), laboratory data and vital sign information.
Statistical methods: The analysis of populations for both the primary and secondary endpoints, the assumptions made within the analyses, and the statistical methods used in the analyses.
Protocol deviations: The determination and evaluation of the potential effect(s) of the analyses on the overall conclusions.
Conclusions: The overall interpretation and assessment of benefits vs. risks associated with the product.
Appendices: The protocol, statistical analysis plan (SAP), case report forms (CRFs) and datasets.[2]

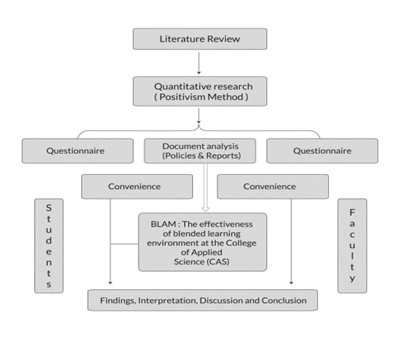
Fig 1 shows the modular structure of a final Clinical study report, detailing core, pre-study, patient data, safety, and statistical sections.
CSR Generation Process
- The process of generating a CSR is organized in a workflow.
- The data must be validated and locked to ensure accuracy.
- The statistical analyses are conducted with the approved SAP.
- The medical writing must be in accordance with ICH E3, as well as with regulatory standards.
- The CSR undergoes quality control and an internal review to ensure consistency and accuracy.
- After the CSR is completed, it will be sent to the regulatory authorities.[3]
Who Prepares CSRs
- Development of pharmaceutical products is conducted by pharmaceutical manufacturers.
- Biotechnology companies conduct clinical trials and conduct R&D.
- Clinical Research Organizations (CROs) support the execution and reporting of clinical trials.
- Medical writing teams develop regulatory submissions.
- Regulatory affairs staff assure compliance with governing laws and regulations.
- Statisticians, clinicians, and data managers work together to guarantee scientific validity and data accuracy.[4]
Value of Clinical Study Reporting
- Required to submit documents and complete the application process.
- Provides assurance that regulatory compliance requirements have been met and data has been properly documented.
- Aids with approvals, labelling, and post-marketing activities.
- Serves as a permanent legal and scientific reference for clinical study.[5]
Simply, A Clinical Study Report (CSR) summarizes the entire study, outlines the methods used during the study, communicates the results of the study, and provides information about safety and efficacy for both patients and regulators.
Transform Complex Clinical Data into Regulatory-Ready Evidence with StatsWork
Reference
- Bellary, S., Krishnankutty, B., & Latha, M. S. (2014). Basics of case report form designing in clinical research. Perspectives in clinical research, 5(4), 159-166. https://journals.lww.com/picp/fulltext/2014/05040/basics_of_case_report_form_designing
- Bucur, M. (2013). The CSR implementation. Acta Marisiensis. Seria Technologica, 10(2), 70. https://amset.umfst.ro/papers/2013-2/12_Bucur_Mihaela%20-%20The_C.S.R._implementation.pdf
- Poolman, R. W., Hanson, B., Marti, R. K., & Bhandari, M. (2007). Conducting a clinical study: A guide for good research practice. Indian Journal of Orthopaedics, 41(1), 27. https://pmc.ncbi.nlm.nih.gov/articles/PMC2981890/
- Glasser, S. P., Salas, M., & Delzell, E. (2007). Importance and challenges of studying marketed drugs: what is a phase IV study? Common clinical research designs, registries, and self‐reporting systems.The Journal of Clinical Pharmacology, 47(9), 1074-1086. https://accp1.onlinelibrary.wiley.com/doi/abs/10.1177/0091270007304776