What are Clinical Research Statistics?
- Home
- Insights
- Article
- What are Clinical Research Statistics?
Qualitative Research Service
News & Trends
Recommended Reads

Data Collection
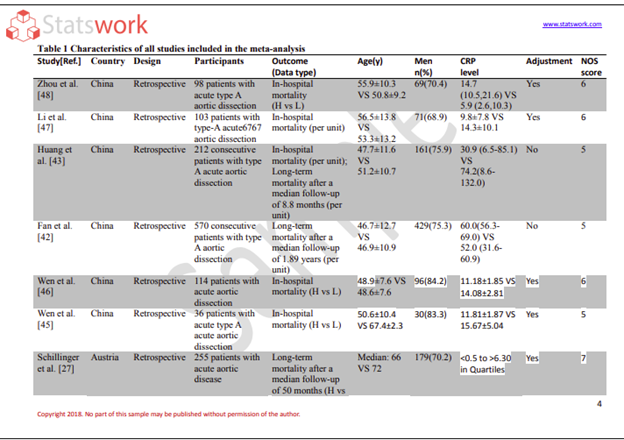
As the data collection methods have extreme influence over the validity of the research outcomes, it is considered as the crucial aspect of the studies
- 1. Introduction
- 2. DeepHealth’s Diagnostic Suite™: Revolutionizing Radiology Workflows
- 3. Key Features
- 4. AI Impact on National Screening Programs
- 5. SmartMammo™: Enhancing Breast Cancer Screening
- 6. DeepHealth AI Use Cases Across Specialties
- 7. Strategic Collaborations and Ecosystem Expansion
- 8. Impact and Adoption of DeepHealth’s AI Solutions
- 9. Conclusion: The Future of Radiology with AI
- 10. References
Statistics in Clinical research means using scientific methods of interpreting data related to healthcare to develop accurate predictions based on statistical analysis of the available data. This includes:
- Evaluate if a treatment really is effective versus appearing to be so only by chance.
- Evaluate who (in addition to the subjects of the study) responded to a specific treatment.
- Provide clinical decision support and frameworks for establishing evidence-based clinical practice guidelines and healthcare policy.[1]
Planning Before You Start
A proper statistical plan will give the structure and direction required to conduct quality, meaningful research. Proper statistical planning helps:
- Sample Size Determination: Identify how many subjects would be required to find a real treatment effect
- Study Design: To select the type of study that would provide the best outcome (e.g., Randomized trials, Cohort, or Case-controlled)
- Outcome Selection: To select the proper measures to accurately measure the success of treatment
- Resource Optimization: To minimize costs and delays while achieving the most reliable results.[2]
Making Sense of Variability
The response of patients to treatments is naturally variable; therefore, clinical research statistics allow researchers to distinguish between:
- True Effects vs Random Differences: Random fluctuations versus truly meaningful results,
- Population Insights: Which groups tend to receive the greatest benefit from a treatment, or are at greater risk, and
- Predictive Analysis: How patients might respond to treatment in the future based on data collected from previous studies.[3]
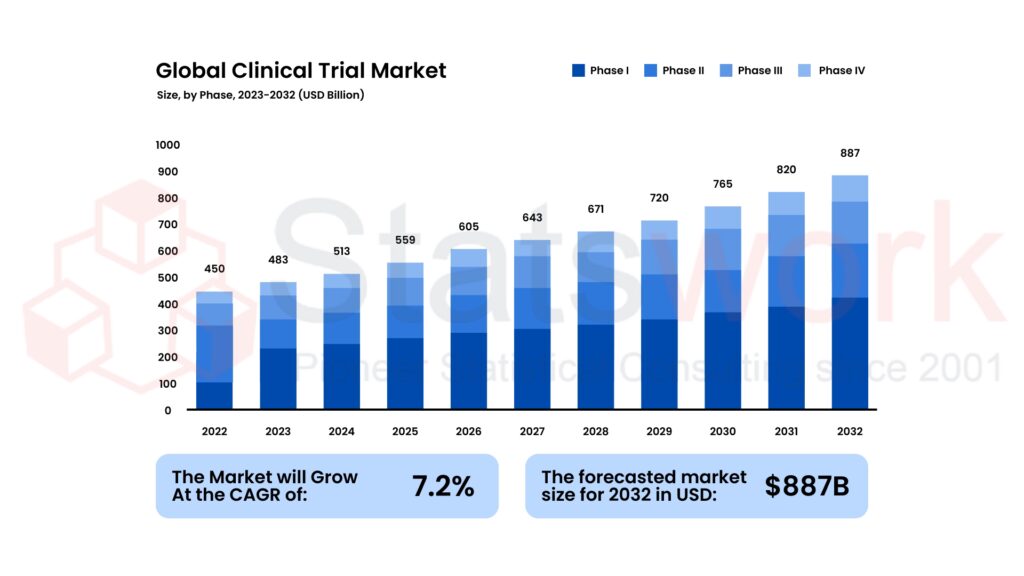
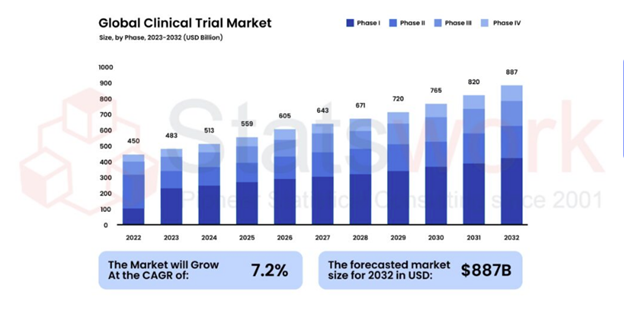
Fig 1 shows the steady global growth of clinical trials across all phases over the forecast period.
Safety and Effectiveness
Researchers must monitor both patient safety (i.e., the degree of safety to which all patients were exposed) and how successful their treatment was. The use of statistical analyses helps researchers too.
- Identify and score on an adverse event and risk occurrence (i.e., an event that might have significant consequences for the patient) early on.
- Evaluate how effective the treatment has been for different patient populations.
- Proactively generate reports that comply with the regulations established for regulatory approval and publication.[4]
From Data to Decisions
The process of converting numbers into actionable insights begins with collecting and analyzing data to develop an appropriate statistical model.
- For doctors, researchers, and policymakers, data should provide evidence-based support for their conclusions.
- Data helps to develop evidence for drug approval, treatment guidelines, and other healthcare-related decisions.
- Data determines the safety and effectiveness of a new medical invention for public use.[4]
Regulatory Compliance and Reporting
In addition to their importance in fulfilling the obligations of law and regulatory compliance, statistics allow Clinical Trial Manufacturers to:
- Generate precise, concise Clinical Study Report contents that contain statistical data.
- Comply with FDA’s, EMA’s, and ICH’s guidelines related to Good Clinical Practice, which are required by law.
- Create data output that is available in auditor/compliance-ready format to enable oversite by regulatory bodies and interested parties.[5]
Thus, the fundamentals of modern medicine rely upon statistically based clinical research. It is through statistical means that clinicians and researchers can analyze large amounts of patient data, turning it into knowledge that they can act on in an evidence-based manner, thereby providing safe, effective, and timely medical advances to patients.
Ensure Accuracy, Compliance, and Impact in Your Clinical Trials — Partner with StatsWork for Expert Biostatistical Analysis.
Reference
- Rodrigues, C. F. D. S., Lima, F. J. C. D., & Barbosa, F. T. (2017). Importance of using basic statistics adequately in clinical research. Revista brasileira de anestesiologia, 67(6), 619-625.https://www.scielo.br/j/rba/a/N5PgBCrzhDkfRbX8QXsctHx/?lang=en
- Gamble, C., Krishan, A., Stocken, D., Lewis, S., Juszczak, E., Doré, C., … & Loder, E. (2017). Guidelines for the content of statistical analysis plans in clinical trials. Jama, 318(23), 2337-2343.https://jamanetwork.com/journals/jama/fullarticle/2666509
- Brennan, P., & Silman, A. (1992). Statistical methods for assessing observer variability in clinical measures. BMJ: British Medical Journal, 304(6840), 1491.https://pmc.ncbi.nlm.nih.gov/articles/PMC1882212/
- Moore, K. L., & van der Laan, M. J. (2009). Application of time-to-event methods in the assessment of safety in clinical trials. In Design and analysis of clinical trials with time-to-event endpoints(pp. 473-500). Chapman and Hall/CRC.https://www.taylorfrancis.com/chapters/edit/10.1201/9781420066401-25/application-time-event-methods-assessment-safety-clinical-trials-kelly-moore-mark-van-der-laan
- Miller, J. E., Korn, D., & Ross, J. S. (2015). Clinical trial registration, reporting, publication and FDAAA compliance: a cross-sectional analysis and ranking of new drugs approved by the FDA in 2012. BMJ open, 5(11), e009758. https://bmjopen.bmj.com/content/5/11/e009758.short