What is Clinical Data Management?
- Home
- Insights
- Article
- What is Clinical Data Management?
Qualitative Research Service
News & Trends
Recommended Reads

Data Collection
As the data collection methods have extreme influence over the validity of the research outcomes, it is considered as the crucial aspect of the studies
- 1. Introduction
- 2. DeepHealth’s Diagnostic Suite™: Revolutionizing Radiology Workflows
- 3. Key Features
- 4. AI Impact on National Screening Programs
- 5. SmartMammo™: Enhancing Breast Cancer Screening
- 6. DeepHealth AI Use Cases Across Specialties
- 7. Strategic Collaborations and Ecosystem Expansion
- 8. Impact and Adoption of DeepHealth’s AI Solutions
- 9. Conclusion: The Future of Radiology with AI
- 10. References
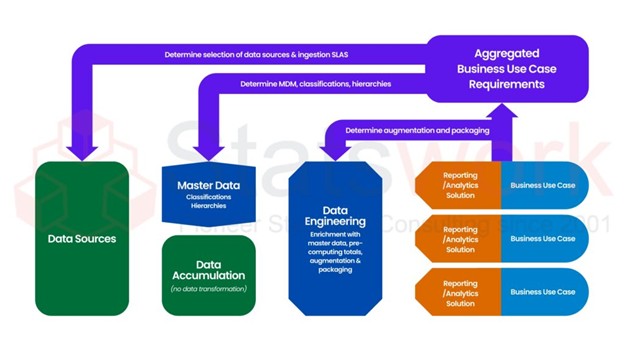
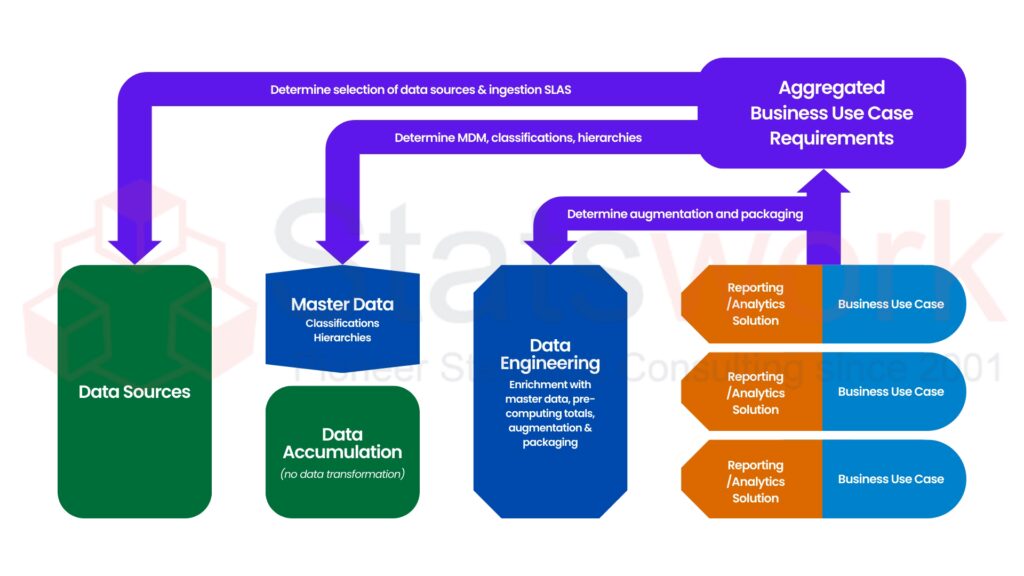
Structured processing needs to be applied for dealing with clinical trial data correctly and effectively. Clinical Data Management (CDM) entails organizing, validating, as well as managing quality clinical data as its primary goal to make it reliable as well as compliant with regulations. Planning, database design, quality management, and secure database lock help CDM achieve reliable analysis, safe drug development, as well as enhanced patient outcomes.[1]
Purpose of Clinical Data Management
- Convert data into clean datasets: Clinical Data Management is involved in the preparation of candidate analysis datasets from raw clinical trial data.
- Collect data from multiple sources: In Clinical Data Management in Clinical Trials, data comes from sources including investigator data, laboratory data, and patient data.
- Review and validate data: The Clinical Data Management Process employs Clinical Data Validation in the process of recognizing discrepancies.
- Maintain quality and compliance: Clinical Data Quality Management and EDC systems ensure data quality.
- Improve efficiency: Clinical Trial Data Management optimizes processes and prevents delays in trials.[2]
Key Components of the CDM Process
Clinical Data Management includes several structured processes that help guide data from inception to ultimate analysis. These components are detailed in the following table:
CDM Component | Description |
Study Setup | Designing CRFs, Data Standards, Validation Rules, and so on. |
Data Collection | Data acquisition through CRFs or Electronic Data Capture (EDC) systems |
Data Validation | Identifying missing, inconsistent, or incorrect data |
Query Management | Communication with Sites to Clarify Discrepancies in Data |
Database Lock | Finalizing clean data for analysis.[3] |
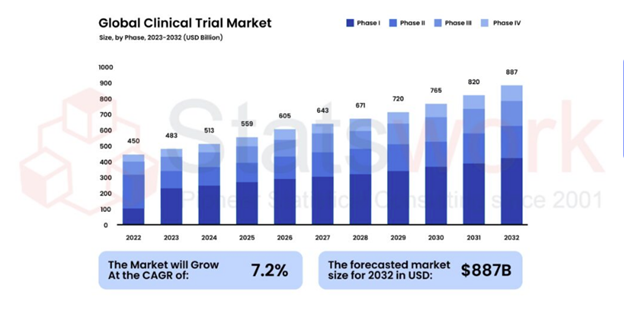
Fig 1 shows the consistent global growth of the Clinical Data Management System market from 2023 to 2033.
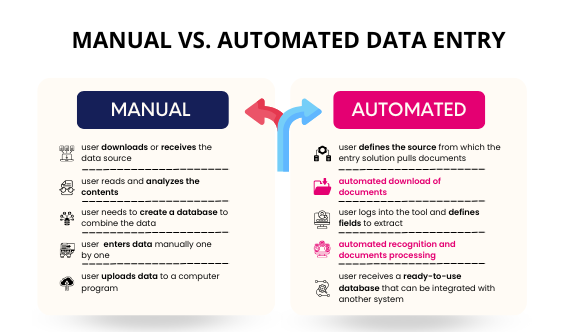
Impact of Digital Technologies on Clinical Data Management
Digital technologies such as EDC solutions, real-time data entry capture, automatic validations, and secure platforms improve accuracy, compliance, teamwork, and overall efficiencies related to clinical data management.
- Electronic Data Capture (EDC) systems: EDC plays a very important role in the data entry process of clinical data management within clinical trials.
- Real-time data entry: EDC streamlines the Clinical Data Management Process by allowing immediate input.
- Automated validation checks: Technology assists Clinical Data Validation through the identification of errors much faster.
- Secure data storage and audit trails: Clinical Data Management Systems guarantee the safety of data and Clinical Data Quality Management.
- Improved collaboration and efficiency: Data management in clinical trials works to allow the enhancement of efficiency between sponsors, CROs, and sites.[2]
Regulatory Compliance and Data Quality in Clinical Data Management
Compliance and data quality for regulatory requirements ensure the integrity and security of data from clinical trials and adherence to regulatory requirements across the globe for trustworthy and ethical research.
- Adherence to global guidelines: The Clinical Data Management in Clinical Trials complies with Good Clinical Practice (GCP), ICH, and other standards and regulations.
- Maintain audit trails: Clinical Data Management Systems allow changes to be tracked to ensure accountability.
- Ensure data integrity: Clinical Data Validation & Quality Checks ensure that data in clinical trials is trustworthy.
- Protect patient confidentiality: Appropriate Clinical Data Management Procedures secure confidential patient data.
- Regulatory acceptance: Effective Clinical Data Management ensures that clinical trial results conform to FDA and EMA regulatory requirements among others.[4]
Importance of Clinical Data Management
- Clinical Data Management provides credible and authentic outcomes for Clinical Trials.
- Effective Clinical Trial Data Management reduces timescales and expenses.
- Correct Clinical Data Management Processes improve decision-making in clinical research.
- Clinical Data Validation and Clinical Data Quality Management guarantee valid data.
- Clinical Data Management plays an integral role in evaluating drug safety and efficacy.[5]
Therefore, Effective Clinical Data Management is a process or system that provides accurate and reliable data of high quality in clinical trials. CDM eliminates inefficiency in data by making use of technological systems and validation.
Unlock reliable clinical trial insights—partner with StatsWork for next-level Clinical Data Management today!
Reference
- Krishnankutty, B., Bellary, S., Kumar, N. B., & Moodahadu, L. S. (2012). Data management in clinical research: an overview. Indian journal of pharmacology, 44(2), 168-172. https://journals.lww.com/iphr/fulltext/2012/44020/data_management_in_clinical_research
- Greenes, R. A., Pappalardo, A. N., Marble, C. W., & Barnett, G. O. (1969). Design and implementation of a clinical data management system. Computers and biomedical research, 2(5), 469-485. https://www.sciencedirect.com/science/article/abs/pii/0010480969900123
- Lu, Z., & Su, J. (2010). Clinical data management: Current status, challenges, and future directions from industry perspectives. Open Access Journal of Clinical Trials, 93-105. https://www.tandfonline.com/doi/abs/10.2147/OAJCT.S8172
- Osifowokan, A. S., & Adukpo, T. K. (2024). The importance of quality assurance in clinical trials: Ensuring data integrity and regulatory compliance in the US pharmaceutical industry. https://www.researchgate.net/profile/Tobias-Adukpo-3/publication/389776596_The_importance_of_quality_assurance_in_clinical_trials
- Gaddale, J. R. (2015). Clinical data acquisition standards harmonization importance and benefits in clinical data management. Perspectives in clinical research, 6(4), 179-183. https://journals.lww.com/picp/fulltext/2015/06040/clinical_data_acquisition_standards