Why Randomized Controlled Trials Remain the Gold Standard in scientific research
- Home
- Insights
- Article
- Why Randomized Controlled Trials Remain the Gold Standard in scientific research
Qualitative Research Service
News & Trends
Recommended Reads

Data Collection
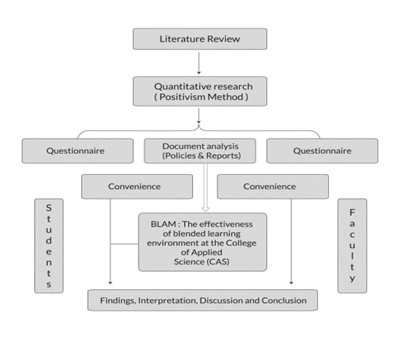
As the data collection methods have extreme influence over the validity of the research outcomes, it is considered as the crucial aspect of the studies
Why Randomized Controlled Trials Remain the Gold Standard in scientific research
- 1. Introduction
- 2. DeepHealth’s Diagnostic Suite™: Revolutionizing Radiology Workflows
- 3. Key Features
- 4. AI Impact on National Screening Programs
- 5. SmartMammo™: Enhancing Breast Cancer Screening
- 6. DeepHealth AI Use Cases Across Specialties
- 7. Strategic Collaborations and Ecosystem Expansion
- 8. Impact and Adoption of DeepHealth’s AI Solutions
- 9. Conclusion: The Future of Radiology with AI
- 10. References
Introduction: Randomized Controlled Trials (RCT)
RCTs are one of the main building blocks of scientific inquiry, particularly when it comes to clinical trial research. The key feature of an RCT is that participants are randomly assigned to treatment groups, resulting in more objective data. Randomization reduces confounding factors and enhances the reliability of clinical trial data.
Another key factor in RCT design is that many clinical trials use double-blind techniques; thus, neither the participants nor the researchers themselves know what treatment group is being administered. This helps to eliminate biased influences on the results of the trial and enhances the reliability of the study results for evaluating medical therapies.[1]
How Randomization Ensures Unbiased Results
- Minimizes Selection Bias: Randomized Controlled Trials (RCT) provide a method of assignment of study subjects into treatment arms that is unbiased.
- Balances Variables: By randomizing participants, RCTs create equal distribution of both known and unknown variables between the treatment groups, which allows researchers to make a more accurate comparison between treatments.
- Increases Internal Validity: Random assignment of subjects allows the equal distribution of both known and unknown factors across all treatment arms, thus enabling a better comparison between the treatments.
- Strengthens Objectivity: The process of randomization permits double blinded clinical trials to be conducted more objectively and gives both the subject and the investigator an equal chance of not knowing which treatment the subject is receiving.[1]
The Importance of Control Groups and Blinding
- The control group enables comparison of the intervention effect compared with a non-treatment group.
- Blinding reduces bias by not allowing participants and researchers to see who has received treatment (also known as double-blinding).
- Blinding controls for the psychological factors of the placebo effect, increasing the confidence that observed changes are related to the intervention and not influenced by other factors.
- Combining the use of control groups with a blinding approach enhances the overall theoretical and empirical credibility of the results produced in clinical studies.[2]
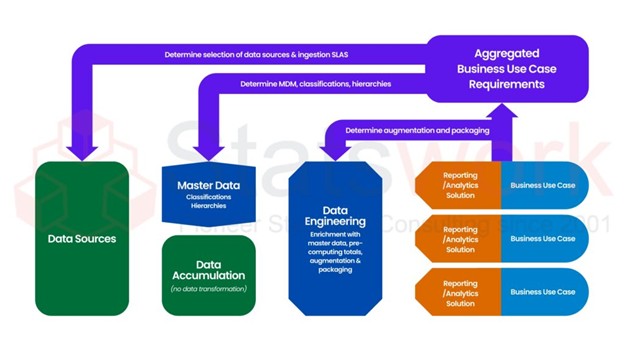
Why Randomized Controlled Trial Superior to Others
Feature | Randomized Controlled Trials | Other Research Methods |
Bias Control | Randomization & Blinding | No randomization or blinding |
Internal Validity | Minimizes confounders | Prone to confounding |
Causality Inference | Can establish cause-effect | Correlation, not causation |
Study Complexity | Requires careful design | Easier to implement |
Ethical Considerations | Challenging for some interventions | Typically, observational. [3] |
Designing an Effective RCT: Best Practices
- A well-designed Randomized Controlled Trial (RCT) requires careful consideration and adherence to the best practices when designing your RCT.
- Critical considerations are randomization of subjects, which eliminates potential bias in clinical research; the use of a control group for comparison; and the use of a double-blind trial for the purpose of minimizing both participant and researcher bias.
- The internal validity of the research must be maintained through a well-structured experimental design, and ethical guidelines must be adhered to for the results of clinical trials and scientific research to be trustworthy and credible.
Ethical Consideration in RCT’s
- Informed Consent: Participants must know about the purpose, risks, and benefits of participating in a Randomized Control Trial (RCT) before they decide voluntarily whether to take part in the study.
- Safety Monitoring: To have protections against possible harm, there should be continuous monitoring throughout the trial, with a plan for ending the trial early if adverse effects are noted.
- Confidentiality: Participant confidentiality will be respected to maintain a participant’s privacy and build trust
- Scientific Integrity: A well-designed experimental research design protocol will result in unbiased research results and will ensure adherence to ethical principles, and it will not allow for the manipulation or fabrication of results.[4]
Role of Statistical Analysis in RCT’s
- Statistical analysis allows you to judge how well your method worked. To do this, you can look at the results of all treatments within an RCT.
- Assist in controlling confounding variables when making comparisons. This includes analysing randomization in clinical research through randomisation and blinding.
- Statistical methods provide stronger validation for findings and reliable conclusions in experimental design. Thus, they enable accurate evaluation of research results.[5]
Challenges in Randomized Controlled Trials (RCT’s)
Recruitment Issues | Insufficient participants may lead to sample bias. |
Ethical Concerns | Participants’ protection, informed consent, and safety should all be addressed when conducting a clinical trial, particularly when it is a high-risk clinical trial. |
Randomization Bias | There is the potential for an imbalanced participant sample if the randomization process is not properly performed. |
Dropouts and Non-compliance | Recruitment may lead to dropouts or non-adherence to the protocol, resulting in impact on the trial results. |
Generalizability | Trial results may not be generalizable to larger populations due to the focus on one specific sample.[2] |
Conclusion
To sum up, RCTs are the best way of doing science because they reduce bias, control for Other Related Issues (bias) & Control for Other Causes (confounding variables) and allow us to determine what causes what (causation).
The best example to illustrate the benefits of RCTs would be conducting properly designed clinical trials (blinding, randomization). RCTs provide accurate data and the most reliable evidence for new treatments and development of medical devices.
Get Unbiased, Reliable Results with RCT-Based Data Collection. Start with Statswork Today!
Reference
- White, H., Sabarwal, S., & De Hoop, T. (2014). Randomized controlled trials (RCTs). Methodological Briefs, Impact Evaluation, 7. https://home.cerge-ei.cz/kaliskova/files/policy_eval/White_Sabarwal_de_Hoop_2014.pdf
- Staudacher, H. M., Irving, P. M., Lomer, M. C., & Whelan, K. (2017). The challenges of control groups, placebos and blinding in clinical trials of dietary interventions. Proceedings of the Nutrition Society, 76(3), 203-212. https://www.cambridge.org/core/journals/proceedings-of-the-nutrition-society/article/challenges-of-control-groups-placebos-and-blinding-in-clinical-trials-of-dietary-interventions/CD0D06FBF583C0E1DE908002A4133335
- Grossman, J., & Mackenzie, F. J. (2005). The randomized controlled trial: gold standard, or merely standard?. Perspectives in biology and medicine, 48(4), 516-534. https://muse.jhu.edu/pub/1/article/188201/summary
- Nicholls, S. G., Carroll, K., Nix, H. P., Li, F., Hey, S. P., Mitchell, S. L., … & Taljaard, M. (2022). Ethical considerations within pragmatic randomized controlled trials in dementia: Results from a literature survey. Alzheimer’s & Dementia: Translational Research & Clinical Interventions, 8(1), e12287. https://alz-journals.onlinelibrary.wiley.com/doi/full/10.1002/trc2.12287
- Faulkner, C., Fidler, F., & Cumming, G. (2008). The value of RCT evidence depends on the quality of statistical analysis. Behaviour Research and Therapy, 46(2), 270-281. https://www.sciencedirect.com/science/article/abs/pii/S0005796707002331