- Services
- Data Analysis services
- Sample Work
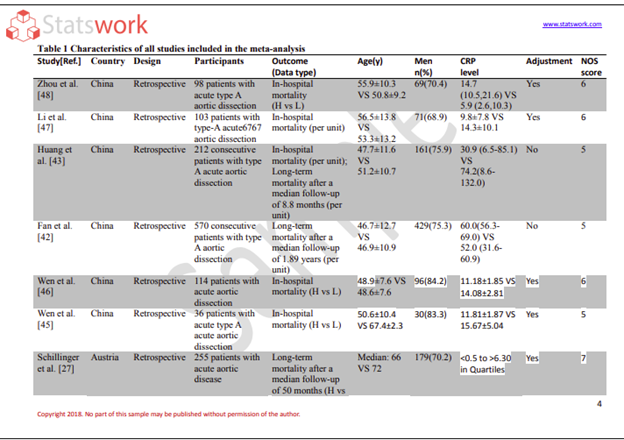
Data Analysis services
- Secondary Qualitative Research Services
- Secondary Quantitative Research Services
- Meta-Analysis Research services
- Sample Work

Meta-Analysis Research Services
- Data Collection Services
- Sample Work
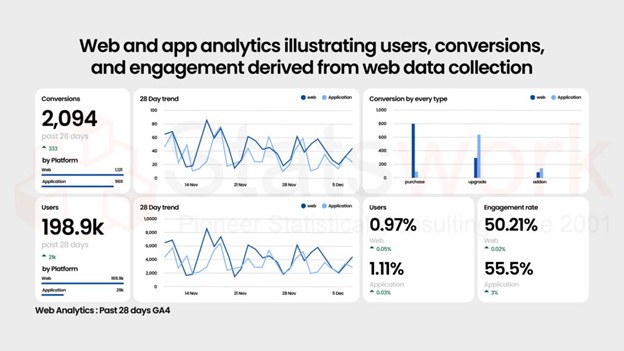
Data Collection Services
- Statistical & Biostatistics services
- Sample Work
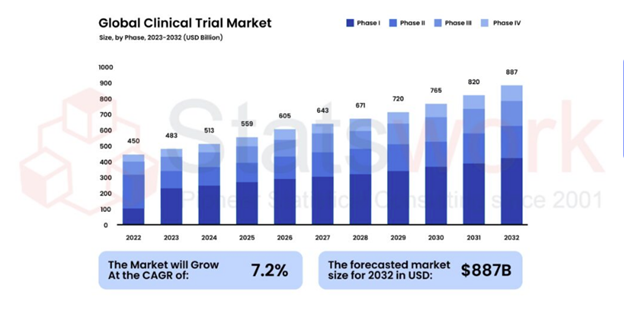
Statistical Programming & Biostatistics services
- Data Management Services
- Sample Work
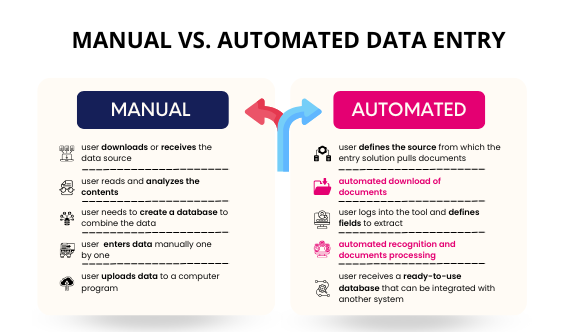
Data Management Services
- Research methodology services
- Sample Work
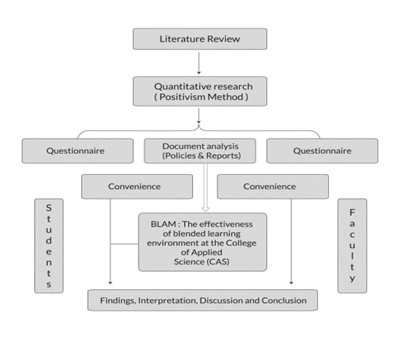
Research methodology services
- Tool Development Services
- Sample Work
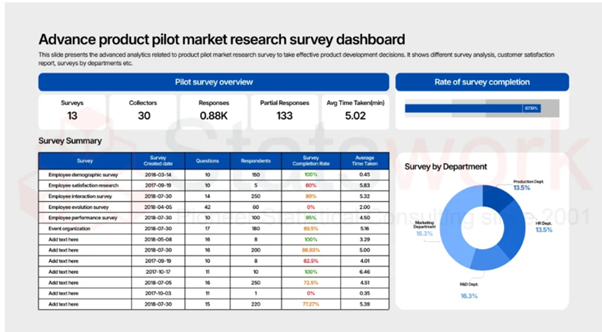
Tool development services
- Statistical Interpretation services
-
Statistical Interpretation services
- Sample Work
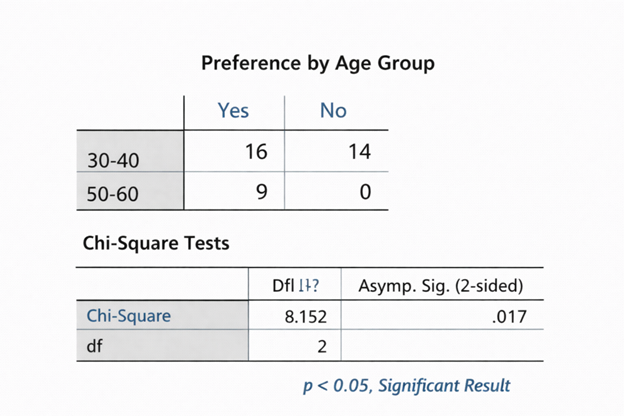
Statistical Interpretation services
-
- Sample Size Calculation Services
-
Sample Size Calculation Services
- Sample Work
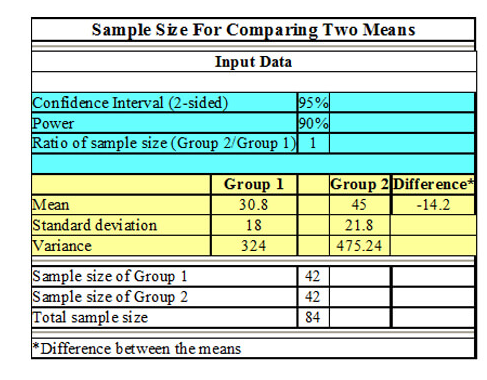
Sample Size Calculation Services
-
- AI & ML Services
-
Artificial Intelligence and Machine Learning Services
- Sample Work
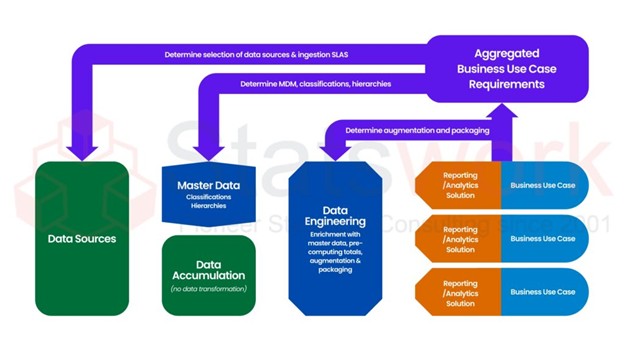
Artificial Intelligence and Machine Learning Services
-
- Meaningful Visualization Services
- Thought Leadership Services
- Report Generation Services
-
Report generation Service
- Sample Work

Report generation Services
-
- Data Analysis services
- Industries
- About Us
- Blog
- Insights
- Contact Us
- Services
- Data Analysis services
- Sample Work

Data Analysis services
- Secondary Qualitative Research Services
- Secondary Quantitative Research Services
- Meta-Analysis Research services
- Sample Work

Meta-Analysis Research Services
- Data Collection Services
- Sample Work

Data Collection Services
- Statistical & Biostatistics services
- Sample Work

Statistical Programming & Biostatistics services
- Data Management Services
- Sample Work

Data Management Services
- Research methodology services
- Sample Work

Research methodology services
- Tool Development Services
- Sample Work

Tool development services
- Statistical Interpretation services
-
Statistical Interpretation services
- Sample Work

Statistical Interpretation services
-
- Sample Size Calculation Services
-
Sample Size Calculation Services
- Sample Work

Sample Size Calculation Services
-
- AI & ML Services
-
Artificial Intelligence and Machine Learning Services
- Sample Work

Artificial Intelligence and Machine Learning Services
-
- Meaningful Visualization Services
- Thought Leadership Services
- Report Generation Services
-
Report generation Service
- Sample Work

Report generation Services
-
- Data Analysis services
- Industries
- About Us
- Blog
- Insights
- Contact Us
Real-World Evidence: Why Pharma Companies Need Advanced Data Analytics in 2025
- Home
- Insights
- Article
- Real-World Evidence: Why Pharma Companies Need Advanced Data Analytics in 2025
Real-World Evidence
- Understanding RWE – Sources and difference from clinical trials
- Advanced Analytics – Turning RWE into actionable insights
- Applications – Drug development, compliance, and market access
- Challenges – Data quality, privacy, and interpretation issues
- Case Study – How Advanced Data Analysis Transformed Pharma Outcomes
- Final Thoughts – The Role of Data Analysis in the Pharmaceutical Industry in 2025
News & Trends
Recommended Reads
Contact us
Real-World Evidence: Why Pharma Companies Need Advanced Data Analytics in 2025
Table of Content
May 2025 | Source: News-Medical
Introduction – Define RWE and its pharmaceutical development implications
Real-World Evidence (RWE) is the set of all information gathered from day-to-day clinical practice via various methods, including electronic health records (EHRs), insurance claims, registries, and other forms of documentation. Through our pharma analytics, RWE analytics, and pharmaceutical research data analysis offerings within the pharmaceutical industry, we support organizations in utilizing that information in a constructive manner so that manufacturers can make informed choices about drug development, which leads to better treatment options for patients and maximized profitability for manufacturers. Our data analysis services in UK enable pharmaceutical companies to process and interpret these datasets efficiently .[1]
Understanding RWE – Sources and difference from clinical trials
Sources for RWE (real-world evidence) Are:
- Patient registries,
- Hospital Records
- observational studies
Clinical trials measure the potential effect of a service in a controlled research environment, whereas RWE collects live data of patients under care for the same disease(s).
By using both RWE and clinical trial data as a combined resource, pharmaceutical manufacturers will be able to determine:
- The effectiveness of therapy
- Patient compliance with treatment regimens
- Long-Term Outcomes.
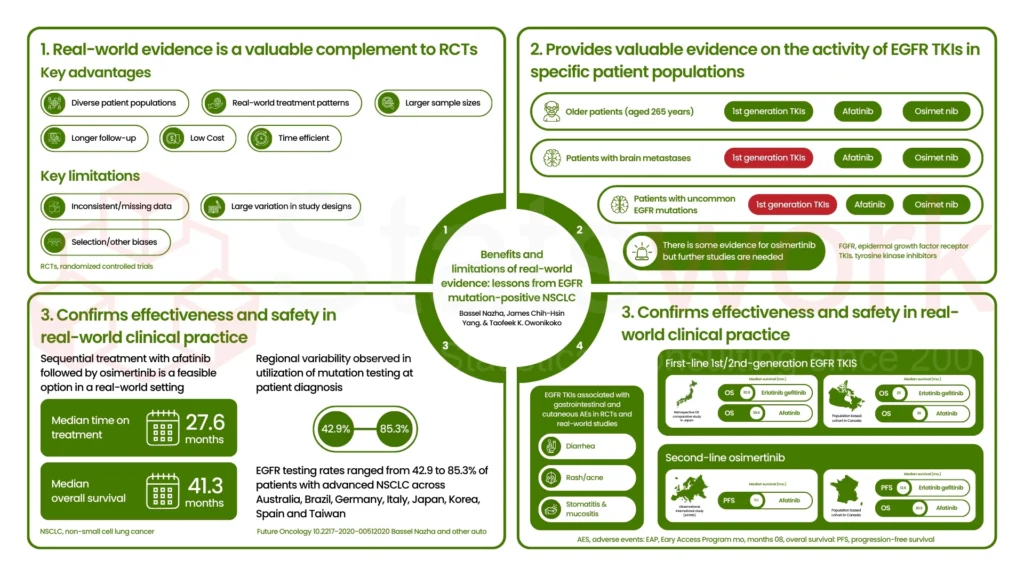
Advanced Analytics – Turning RWE into actionable insights
Aspect | Short Description | Examples |
Advanced Analytics | Derives important information out of RWD. | Determine optimal drug dosage based on patient data. |
Healthcare Analytics | Identifies trends and patterns within RWD. | Adverse Events Dashboard tracking RWD adverse events. |
Predictive Modeling | Predicting patient outcomes and treatment responses. | Predict patients with high risk for readmission.[2] |
Applications – Drug development, compliance, and market access
- Pharmaceutical Applications – Drug Development, Compliance, and Access to Market Data analysis within the pharmaceutical industry plays an important part in
- Drug development: RWE analytic tools will help optimize trial design, progress monitoring and the identification of patient populations.
- Regulatory Compliance: support for submission and safety monitoring.
- Market Access: improving pricing and reimbursement decision-making and facilitating better engagement with relevant stakeholders. [3]
Data-driven strategies and Improved Patient Outcomes via A Pharma Research Data/RWE Analytics.
Challenges – Data quality, privacy, and interpretation issues
RWE is an important source of information. However, there are challenges associated with inconsistent data quality, difficulty integrating many different types of data, and complying with various privacy regulations. To obtain reliable evidence from pharmaceutical data, a strong healthcare analytics approach to engaged clinical data is critical, offered by leading data analytics companies UK.
Aspect | Aspect of Extensive Data Use |
2025 Pharma | Focus Growing dependence on data analysis, real-world evidence analytics, and AI-enabled solutions as competition increases. |
Emerging Trend 1 | Predictive Modeling for Patient Outcome Forecasting |
Emerging Trend 2 | AI-Powered Trial Simulations Enhance Study Design and Cost Reduction [4] |
Emerging Trend 3 | Real-Time Monitoring to Support Immediate and Accurate Decision Making |
Advantage | Adoption of These Technologies Today Provides Immediate Insight, Increase Efficiency in Drug Development, and Better Market Performance.[5] |
Example – A pharma company uses AI-driven RWE analytics to predict patient outcomes, run virtual trial simulations, and monitor real-time safety data—leading to faster decisions and more efficient drug development.
Case Study – How Advanced Data Analysis Transformed Pharma Outcomes
Category | Description | Metric |
Data Digitization | data digitised and collected from machines for AI analysis | 90% manual entry has been replaced |
Optimizing Machinery | improving the efficiency of equipment by utilising AI | 25% increase in equipment uptime |
Machine Failure Analysis | ai can detect machine failure patterns and trends to help prevent downtime [3] | 30% decrease in unscheduled downtime |
Smarter Procurement Guidance | AI provides guidance on purchasing and inventory decisions | 15% decrease in procurement costs |
Increased Operational Efficiency | AI helps streamline operational processes and make good decisions – | 20% increase in operational efficiency |
Final Thoughts – The Role of Data Analysis in the Pharmaceutical Industry in 2025
By 2025, strong data analysis will be required if pharmaceutical (pharma) companies intend to be competitive with their competition. Each of these will have RWE analytics, Pharma analytics and their use of artificial intelligence (AI) to be able to derive insights from data.[5] Using strong, quality pharmaceutical research data and the latest healthcare analytics would allow these companies to develop drugs effectively, support regulatory compliance and make quicker and smarter decisions. Advanced use of clinical data analysis and Drug Development analytics will lead to improved results with lower risk and prepare these companies for the continued evolution of the pharmaceutical industry.
Call to Action (CTA)
Unlock the full potential of your pharma data with Statswork.
From RWE analytics to clinical data interpretation, our expert-driven data analysis solutions help you make confident, faster, and more impactful decisions.
- Partner with Statswork for accurate, insight-driven Data Analysis today.
Contact us now to transform your pharma data into measurable results.
References
- Lamberti, M. J., Kubick, W., Awatin, J., McCormick, J., Carroll, J., & Getz, K. (2018). The use of real-world evidence and data in clinical research and postapproval safety studies. Therapeutic Innovation & Regulatory Science, 52(6), 778-783.https://journals.sagepub.com/doi/abs/10.1177/2168479018764662
- Bashirynejad, M., Soleymani, F., Nikfar, S., Kebriaeezadeh, A., Majdzadeh, R., Fatemi, B., … & Zare, N. (2024). Trends analysis and future study of the pharmaceutical industry field: a scoping review. DARU Journal of Pharmaceutical Sciences, 33(1), 6.https://link.springer.com/article/10.1007/s40199-024-00550-x
- Berger, M. L., Ganz, P. A., Zou, K. H., & Greenfield, S. (2024). When will real-world data fulfill its promise to provide timely insights in oncology?. JCO Clinical Cancer Informatics, 8, e2400039.https://ascopubs.org/doi/full/10.1200/CCI.24.00039
- Krendyukov, A. (2025). Navigating an Evolving Landscape in the Pharmaceutical Industry. In Medical Affairs’ Fundamentals: The Next Generation(pp. 17-37). Cham: Springer Nature Switzerland.https://link.springer.com/chapter/10.1007/978-3-031-92588-7_2
- Bodiwala, K., Lalwani, R., Jain, Z., & Gajjar, A. (2025). Data Science for Analytical Development and Quality Control. Data Science in Pharmaceutical Development, 337-383.https://onlinelibrary.wiley.com/doi/abs/10.1002/9781394287383.ch11
