- Services
- Data Analysis services
- Sample Work
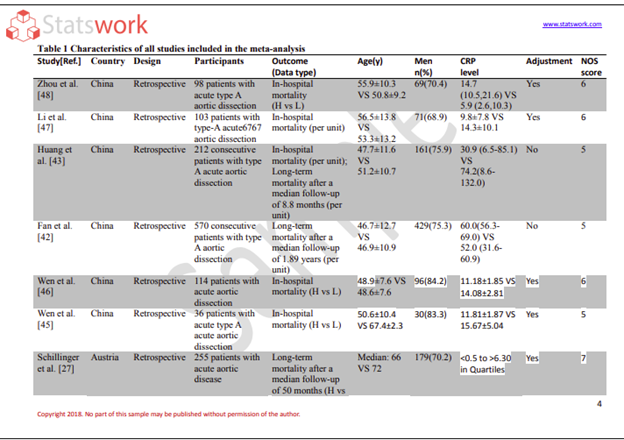
Data Analysis services
- Secondary Qualitative Research Services
- Secondary Quantitative Research Services
- Meta-Analysis Research services
- Sample Work

Meta-Analysis Research Services
- Data Collection Services
- Sample Work
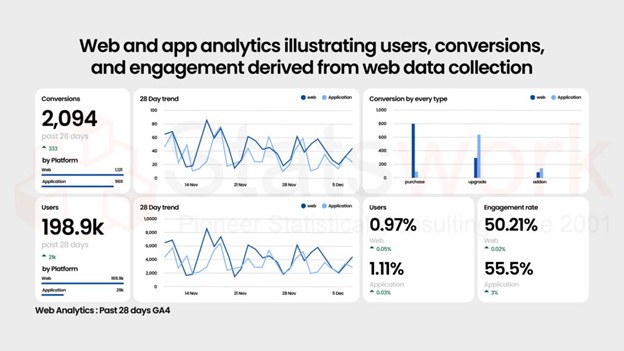
Data Collection Services
- Statistical & Biostatistics services
- Sample Work
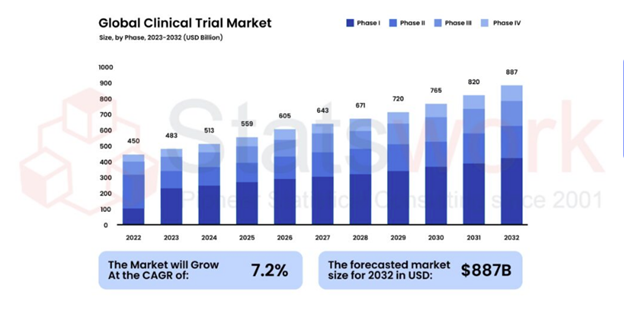
Statistical Programming & Biostatistics services
- Data Management Services
- Sample Work
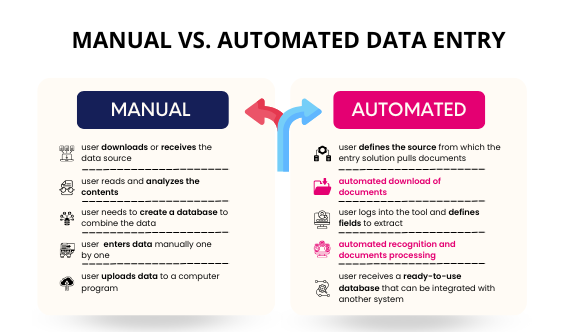
Data Management Services
- Research methodology services
- Sample Work
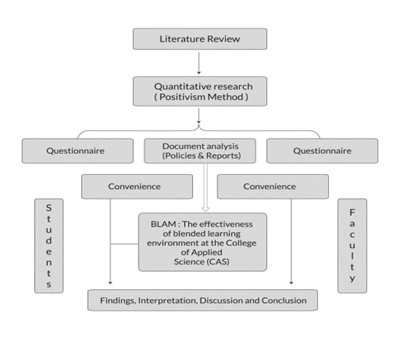
Research methodology services
- Tool Development Services
- Sample Work
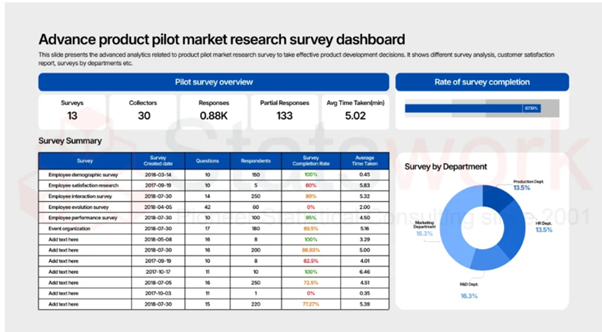
Tool development services
- Statistical Interpretation services
-
Statistical Interpretation services
- Sample Work
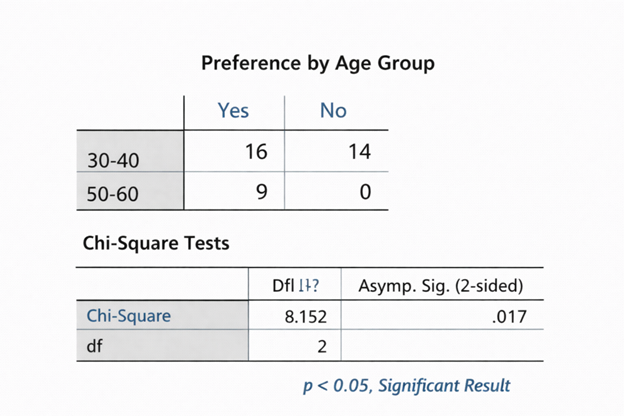
Statistical Interpretation services
-
- Sample Size Calculation Services
-
Sample Size Calculation Services
- Sample Work
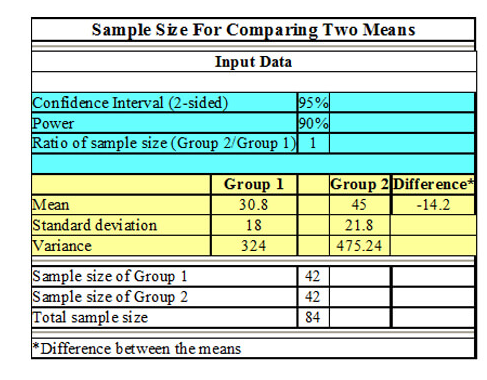
Sample Size Calculation Services
-
- AI & ML Services
-
Artificial Intelligence and Machine Learning Services
- Sample Work
Artificial Intelligence and Machine Learning Services
-
- Meaningful Visualization Services
- Thought Leadership Services
- Report Generation Services
-
Report generation Service
- Sample Work

Report generation Services
-
- Data Analysis services
- Industries
- About Us
- Insights
- Blog
- Contact Us
- Services
- Data Analysis services
- Sample Work

Data Analysis services
- Secondary Qualitative Research Services
- Secondary Quantitative Research Services
- Meta-Analysis Research services
- Sample Work

Meta-Analysis Research Services
- Data Collection Services
- Sample Work

Data Collection Services
- Statistical & Biostatistics services
- Sample Work

Statistical Programming & Biostatistics services
- Data Management Services
- Sample Work

Data Management Services
- Research methodology services
- Sample Work

Research methodology services
- Tool Development Services
- Sample Work

Tool development services
- Statistical Interpretation services
-
Statistical Interpretation services
- Sample Work

Statistical Interpretation services
-
- Sample Size Calculation Services
-
Sample Size Calculation Services
- Sample Work

Sample Size Calculation Services
-
- AI & ML Services
-
Artificial Intelligence and Machine Learning Services
- Sample Work

Artificial Intelligence and Machine Learning Services
-
- Meaningful Visualization Services
- Thought Leadership Services
- Report Generation Services
-
Report generation Service
- Sample Work

Report generation Services
-
- Data Analysis services
- Industries
- About Us
- Insights
- Blog
- Contact Us
How to Design and Analyze Clinical Trials: A Complete Guide
- Home
- Blog
- How to Design and Analyze Clinical Trials: A Complete Guide
Qualitative Research Service
- What Are the Key Fundamentals of Clinical Trials?
- How to Choose the Right Clinical Trial Design and Why It’s Important
- How to Determine Sample Size and Why Statistical Power Matters
- What Makes a Regulatory-Compliant Clinical Trial Protocol?
- How to Plan Data Management and Statistical Analysis Effectively
- How to Interpret Results and Avoid Common Clinical Trial Mistakes
Recommended Reads
Contact us
Introduction: What Is Clinical Trial Design and Why It Matters
- 1. What Are the Key Fundamentals of Clinical Trials?
- 2. How to Choose the Right Clinical Trial Design and Why It’s Important
- 3. How to Determine Sample Size and Why Statistical Power Matters
- 4. What Makes a Regulatory-Compliant Clinical Trial Protocol?
- 5. How to Plan Data Management and Statistical Analysis Effectively
- 6. How to Interpret Results and Avoid Common Clinical Trial Mistakes
The work done to prepare for running a clinical trial that runs smoothly is called “clinical trial design.” To perform well on clinical analysis, there must be a well-designed study done. People will often run their clinical studies using a design with an experimental vs. control group and minimize the chance of bias by using a placebo [1].
Knowledge of how to run a clinical study in a stepwise fashion, use the appropriate biostatistics and meet the regulatory requirements of your country’s clinical research agency, is critical for success. The clinical trial statistical analysis roadmap combined with proper clinical research data management and clinical data analysis will yield scientifically valid and regulatory-compliant results.
What Are the Key Fundamentals of Clinical Trials?
In clinical trial design, it is vital to have clearly defined objectives so that you can conduct an accurate analysis of your clinical trial.
- Having clearly defined eligibility criteria and study population further strengthens the methodology of your clinical trial.
- Most clinical trials are conducted using a randomized controlled method. This typically involves a placebo using a control group to help eliminate bias.
- When understanding the process of designing and conducting a clinical trial step-by-step, this will allow for a structured approach with reliable results.
- Using proper biostatistics to determine the statistical power of your sample size and to assist in analyzing your clinical data is necessary for producing valid results [2].
- The importance of maintaining compliance with applicable regulatory requirements is to assure the protection of patients and the adherence to global regulations.
- To produce quality analysis, an accurate clinical research data management system is necessary to conduct high-quality analyses on your clinical data.
- A well-defined clinical trial statistical analysis guide will clearly outline the statistical method used to analyze your data prior to evaluating the data [3].
How to Choose the Right Clinical Trial Design and Why It’s Important
Choosing the right Clinical Trial Design is critical because it directly affects data quality, regulatory approval, timelines, cost, and the validity of your Clinical Trial Analysis. Below is a clear comparison table to help guide decision-making.
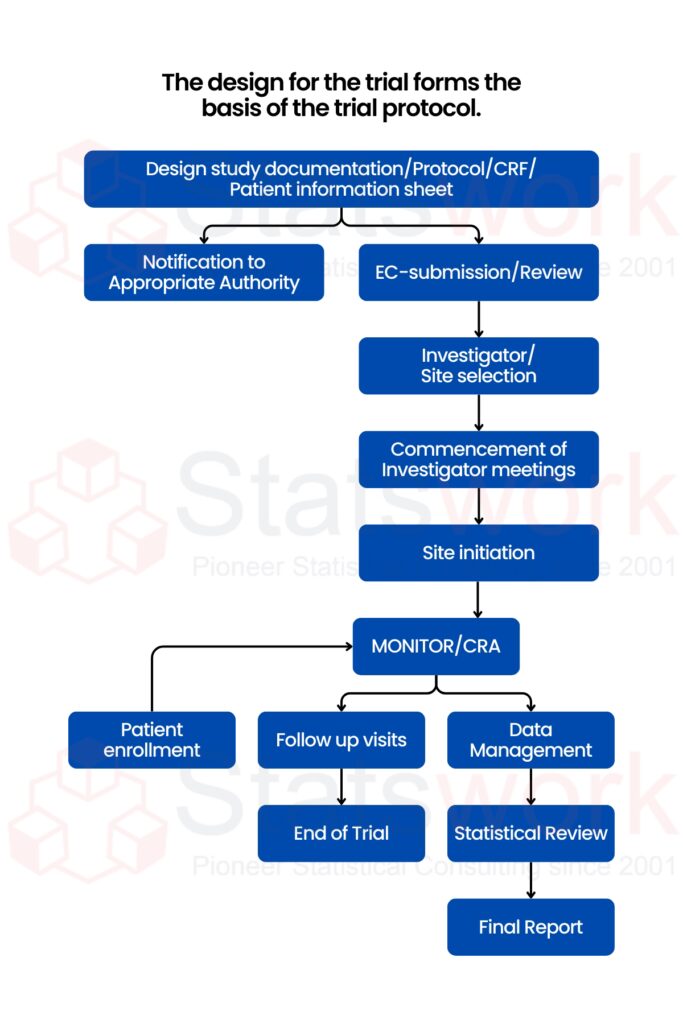
Fig 1. Clinical Trial Design workflow from protocol development to statistical review and final report.
Clinical Trial Design Selection Guide
Design Type | When to Use | Importance | Key Considerations |
Randomized Controlled Trials (RCTs) | When comparing new treatments to standard therapy | Minimizes bias, strengthens analysis of clinical trial data | Requires proper randomization, control group, and sometimes a placebo. |
Parallel Design | When testing two or more treatments at the same time | Easy structure; easy statistical comparison. | Adequate sample size and strong biostatistics plan are required. |
Crossover Design | When subjects can receive multiple treatments over time | Reduces variability of subjects acting as their own control group. | Not appropriate for treatments with long-term effects. |
Adaptive Design | When flexibility is required in the trial | Allows modification of trial based on interim results; maintains regulatory compliance [4]. | Requires advanced biostatistics; pre-defined statistical rules. |
Observational Studies | When randomization is not possible | Good for analyzing actual clinical instances of data. | The risk of bias is higher than in randomized controlled trials. |
How to Determine Sample Size and Why Statistical Power Matters
Sample size is an important aspect of Clinical Trial Design because it helps to determine the reliability of Clinical Trial Analysis. The use of biostatistics helps to ensure that there is sufficient statistical power to identify actual treatment effects while staying within regulatory requirements.
- Define Endpoints Clearly – Primary outcomes help to determine sample size in any trial design.
- Estimate Effect Size – Differences in randomized controlled trials, including control group or placebo comparisons, help to determine sample size.
- Set Power and Significance Levels – Biostatistics (usually 80-90% power) help to ensure reliable clinical data analysis.
- Adjust for Dropouts – Variability helps to ensure that the study remains valid.
- Use a Clinical Trial Statistical Analysis Guide – Planning and clinical research data management help to ensure overall results [5].
Knowing how to design clinical trials helps to ensure that the results are scientifically valid and statistically sound.
What Constitutes a Regulatory-Compliant Clinical Trial Protocol?
A regulatory-compliant protocol in Clinical Trial Design is one that clearly defines trial methodology, such as randomized controlled trials with a control group or placebo and is in regulatory compliance.
It integrates biostatistics, a clinical trial statistical analysis guide, and clinical research data management to ensure proper Clinical Trial Analysis and clinical data analysis.
Understanding the design of clinical trials step by step ensures that the results are ethical, scientific, and regulatory compliant
How to Plan Data Management and Statistical Analysis Effectively
Component | Reason for Use |
Clinical Research Data Management | Will ensure that data collected is accurate and complete to enable trustworthy clinical trial analysis based on the Clinical Trial Design. |
Clear Trial Methodology | Will allow endpoints to be defined along with structure of randomized controlled trials (control group/placebo), to ensure regulatory compliance [2]. |
Biostatistics Planning | Will identify needed sample sizes, necessary power, and assist with trustworthy clinical data analysis. |
Clinical Trial Statistical Analysis Guide | Will define methods of analysis prior to conducting analysis to provide unbiased and transparent analysis results. |
Step by Step Approach | Will ensure consistency in clinical trial design/compliance with regulatory standards through adherence to a step-by-step method to design clinical trials. |
Effective planning strengthens scientific validity and improves overall study success.
How to Interpret Results and Avoid Common Clinical Trial Mistakes
Result interpretation is an important aspect of Clinical Trial Analysis. Effective Clinical Trial Design and biostatistics help in making sure that the results are not only statistically significant but also clinically relevant and compliant with regulatory requirements.
How to Interpret Results Effectively
- Compare Statistical vs. Clinical Significance – Results should be statistically significant in clinical data interpretation but also demonstrate practical patient benefit.
- Examine Confidence Intervals and Effect Size – More informative than p-values in randomized controlled clinical trials.
- Compare Control Group and Placebo Results – Make sure that the results are a true measure of treatment effect within the clinical trial framework.
- Adhere to the Clinical Trial Statistical Analysis Guide – Follow established guidelines to ensure that Clinical Trial Analysis is unbiased.
Common Clinical Trial Errors to Avoid
- Underpowered Clinical Trials – Biostatistics planning in clinical trials can compromise results.
- Protocol Violations – Not following established guidelines on how to design clinical trials can compromise results.
- Inadequate Clinical Research Data Management – Inaccurate data can lead to erroneous clinical data analysis [3].
- Disregard for Regulatory Compliance – Can cause delays or even render clinical trial results null and void.
Conclusion: What Should You Do Next and How to Move Forward Successfully
To move forward successfully, you should review your Clinical Trial Design and make sure that your trial design, including randomized controlled trials with a control group or placebo, meets the standards for regulatory compliance.
You should enhance your biostatistics planning, update your clinical trial statistical analysis guide, and optimize clinical research data management to facilitate correct Clinical Trial Analysis and clinical data analysis [5].
Knowing how to design clinical trials step by step will ensure that you have successful and scientifically valid results.
Reference:
- Armitage, P. (1996). 1 The design and analysis of clinical trials. Handbook of Statistics, 13, 1-29.https://www.sciencedirect.com/science/chapter/handbook/abs/pii/S0169716196130030
- Furberg, C. D. (2010). How should one analyse and interpret clinical trials in which patients don’t take the treatments assigned to them? Journal of the Royal Society of Medicine, 103(5), 202-204.https://journals.sagepub.com/doi/full/10.1258/jrsm.2009.09k074
- Pocock, S. J. (1997). Clinical trials with multiple outcomes: a statistical perspective on their design, analysis, and interpretation. Controlled clinical trials, 18(6), 530-545.https://www.sciencedirect.com/science/article/abs/pii/S0197245697000081
- Jacobe, H. T., Leitenberger, J. J., & Bergstresser, P. R. (2007). Understanding clinical trial outcomes: design, analysis, and interpretation. Dermatologic therapy, 20(2), 77-85.https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1529-8019.2007.00115.x
- Kianifard, F., & Islam, M. Z. (2011). A guide to the design and analysis of small clinical studies. Pharmaceutical Statistics, 10(4), 363-368.https://onlinelibrary.wiley.com/doi/abs/10.1002/pst.477
