Q & A
Statistical Programming & Biostatistics
Q2. How does biostatistics support clinical and healthcare research?

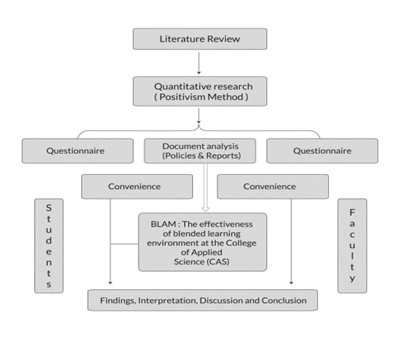
The role of biostatistics in research, specifically during the research process, is to take unprocessed medical data and turn it into scientific evidence that will enable us to make informed decisions within clinical and healthcare research.
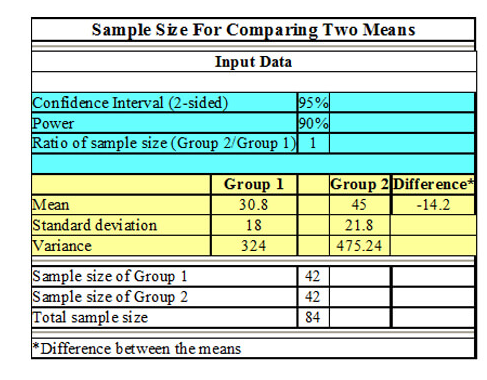
1.Study Design and Planning
Allow for appropriate selection of methodologies, defining of primary/secondary endpoints, estimation of sample size (power analysis), as well as designing clinically and observationally robust studies. (i.e., Efficient study, conducted ethically, Detect true treatment effects.)
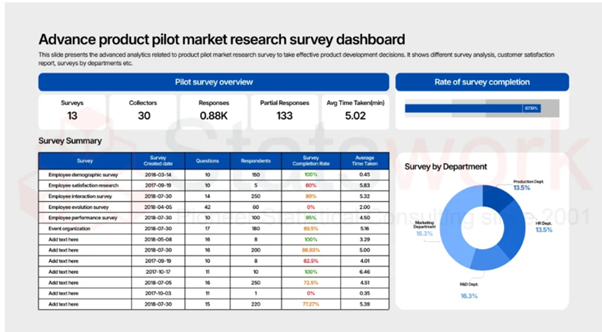
2. Data Quality and Management
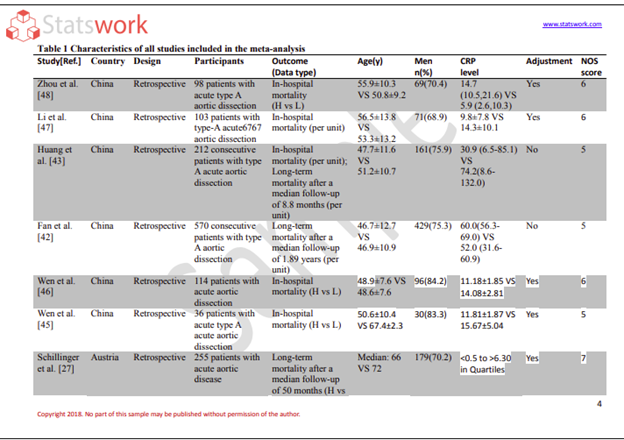
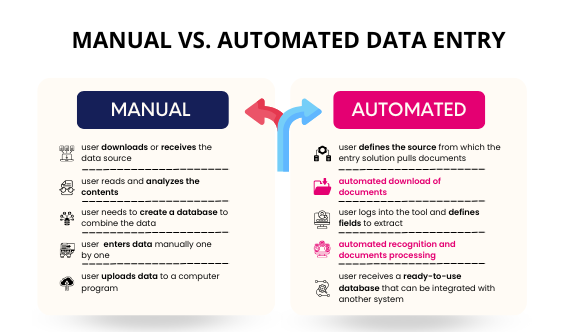
Conduct standardized processes for data collection, validation and management. The analytical methods employed in biostatistics effectively deal with the problems of missing data, measurement errors, and outliers to provide a complete, accurate dataset suitable for analysis.
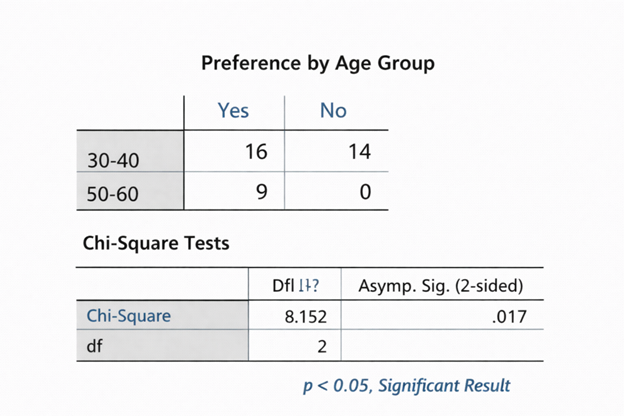
3.Statistical Analysis of Clinical Data
Allows for rigorous analysis of data utilizing methodologies such as regression model(s), survival analysis, longitudinal analyse to evaluate efficacy/safety of treatment, the progression of disease, and risk factors.
4. Regulatory and Ethical Compliance
Support all clinical trials and healthcare studies comply with regulatory guidelines established by governing agencies (FDA, EMA, and ICH). This provides regulatory authorities proper statistical justification when we submit efficacy studies and ethical approvals.
5. Interpretation and Evidence-Based Decisions
Provide clear understanding of complex results through tables, reports and visual depictions of results that enable clinicians, researchers, and decision makers to make informed, evidence-based decisions that lead to improved quality of care for their patients.